For example night-vision goggles operate by detecting infrared light as shown in Figure 9.2.2. While people cannot see this light, some animals can and we can make cameras that can detect this light. This light is called infrared, because its frequency is closest to red in visible light. For visible light the charges have to be oscillating back-and-forth around 10 14 times per second. For objects at room temperature (around 300K) the oscillating atoms give off electromagnetic light at roughly 10 13 Hz. Except the light created in these circumstances is not visible light. In fact we do give off electromagnetic radiation as we sway back and forth, and objects in dark rooms do "glow". In fact, it seems if light is the oscillation of charge it should be very difficult to find darkness at all. By swaying backwards and forwards we were making those charges oscillate, but we did not seem to suddenly create light. In Physics 7A we learned that atoms at temperatures higher than absolute zero oscillated, and these atoms are made up of electrons and protons, yet most objects do not appear to glow in the dark. Most of us have been zapped by a sweater we were wearing at some point in our lives, due to the build-up of charge on it. The frequency with which charges oscillate up and down sets the frequency of the electromagnetic waves produced, like similar to how the frequency of the wave on a string is set by a person at the end of the string, oscillating it.įigure 9.2.2: Electromagnetic Spectrum of Lightįor electromagnetic waves, like all the other waves we have studied, the frequency is determined by the source. Below is an image of the electromagnetic spectrum (the meaning of this term will become more useful in later chapters). As we will explore in greater detail in later chapters, the sources of electromagnetic waves (i.e. In fact, we categorize light of different frequencies into groups due to its various properties that arise. The light that we experience every day is made up of many photons in a range of frequencies, so we don’t notice the quantized nature of light any more than we notice the individual atoms in everyday materials. So the smallest amount of energy in light is different for each type of light. We just concluded that each photon is an indivisible quantum of light, but photons of different frequencies contain different amounts of energy. 
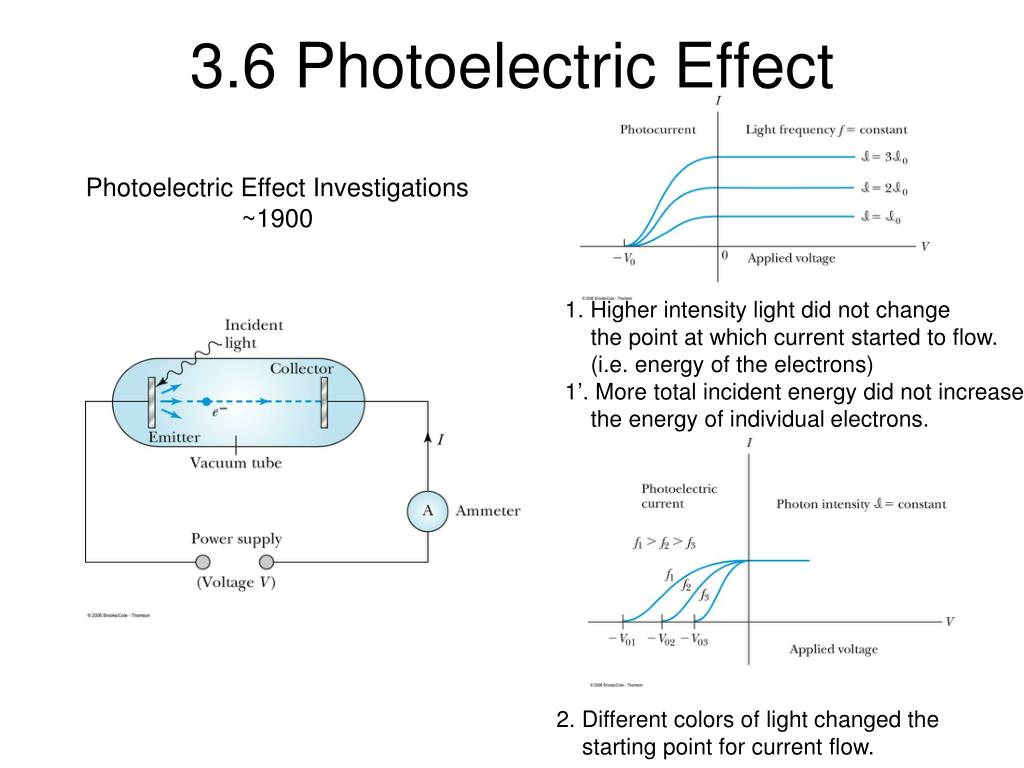
Again, the models make different predictions. In the wave model, a greater brightness would indicate a larger amplitude wave we would conclude that greater intensity waves have the ability to transfer larger amounts of energy into another system. Using the particle model, we conclude that the brightness of the beam does not influence how much energy any particular photon can transfer to another system. Thus, more electrons are ejected, but each ejected electron still has the same kinetic energy. 
In the particle model, we would describe this as adding more photons to the beam, but each particular photon still only carries a certain amount of energy. Next, consider the action of increasing the beam's intensity. Clearly, the two models lead to different hypotheses. The energy in a wave is related to its amplitude, so it would seem both light beams must have equal ability to transfer energy. But in the wave model, the same intensity of each beam means they must have the same amplitude. Classical physics tried to explain the photoelectric effect by justifying the electron emission from a light-irradiated metal target by the thermal emission which occurs when a piece of metal is heated to high temperatures. This tells us that the beam with the higher frequency has fewer photons. But if the intensities of the beams are same, the total energy transferred by each beam is the same. Thus, the high frequency beam is capable of transferring larger amounts energy into another system. The lower the energy the light, the less work can be done with it by the atom when it absorbs that light.\( \newcommand, we conclude that the beam with the higher frequency has photons with higher energy. Radio waves cause nuclear spin transitions which is when a proton changes its spin state. Microwaves cause rotational motion where a molecule rotates. The bonding atoms of a molecule vibrate back and forth like an oscillating spring. Infrared light causes molecular vibrations. The electrons are able to move between the energy levels within the atom, but do not have enough energy to escape. Low energy UV and visible light cause electron transitions. They transfer enough energy to electrons so they can escape from the pull of the atom’s nucleus and turn the atom into an ion. Higher energy light such as gamma rays, X-rays, and high energy UV light cause ionizations. And since energy and frequency are directly proportional, the trend we describe using energy will be the same for frequency. The different effects light has on atoms can best be understood when considering the energies of types of light.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
